What Counts as “Research” on Mad Honey?
Before you interpret any claim, it helps to know what the “evidence pile” actually contains. Most people imagine research means large randomized trials with clear benefits and recommended doses. For mad honey, that’s not the reality.
The research landscape is more fragmented: some clinical case literature, some animal experiments, and a set of analytical chemistry papers that focus on identifying the active compounds.
Human evidence: case reports and case series
The most common human “research” is not a trial. It’s clinicians documenting what happens when people show up at the hospital after consuming mad honey. These papers are valuable because they show repeatable symptom patterns, typical time course, and what medical teams monitor, but they don’t demonstrate a “benefit” in healthy people.
Animal studies: signals, not proof for humans
Animal studies can explore mechanisms, dose-response relationships, and biological markers in controlled settings. That’s useful for learning what might be happening, but it does not automatically mean “this will happen in humans,” especially when studies use extracts, concentrates, or doses that don’t reflect normal consumption.
Lab/chemistry research: what’s in it and how it’s measured
Analytical chemistry papers are often the most concrete part of the research ecosystem because they deal with measurable realities: identifying and quantifying grayanotoxins and checking for contaminants or adulteration markers. This is where the conversation becomes less hype and more methodology.
Why you rarely see large controlled human trials
There are a few practical reasons: inconsistent natural batches, ethical limits around deliberately inducing adverse effects, regulatory complexity, and the lack of a standardized “active dose” across products. When a product varies jar-to-jar, it’s much harder to design clean trials that tell you anything definitive.
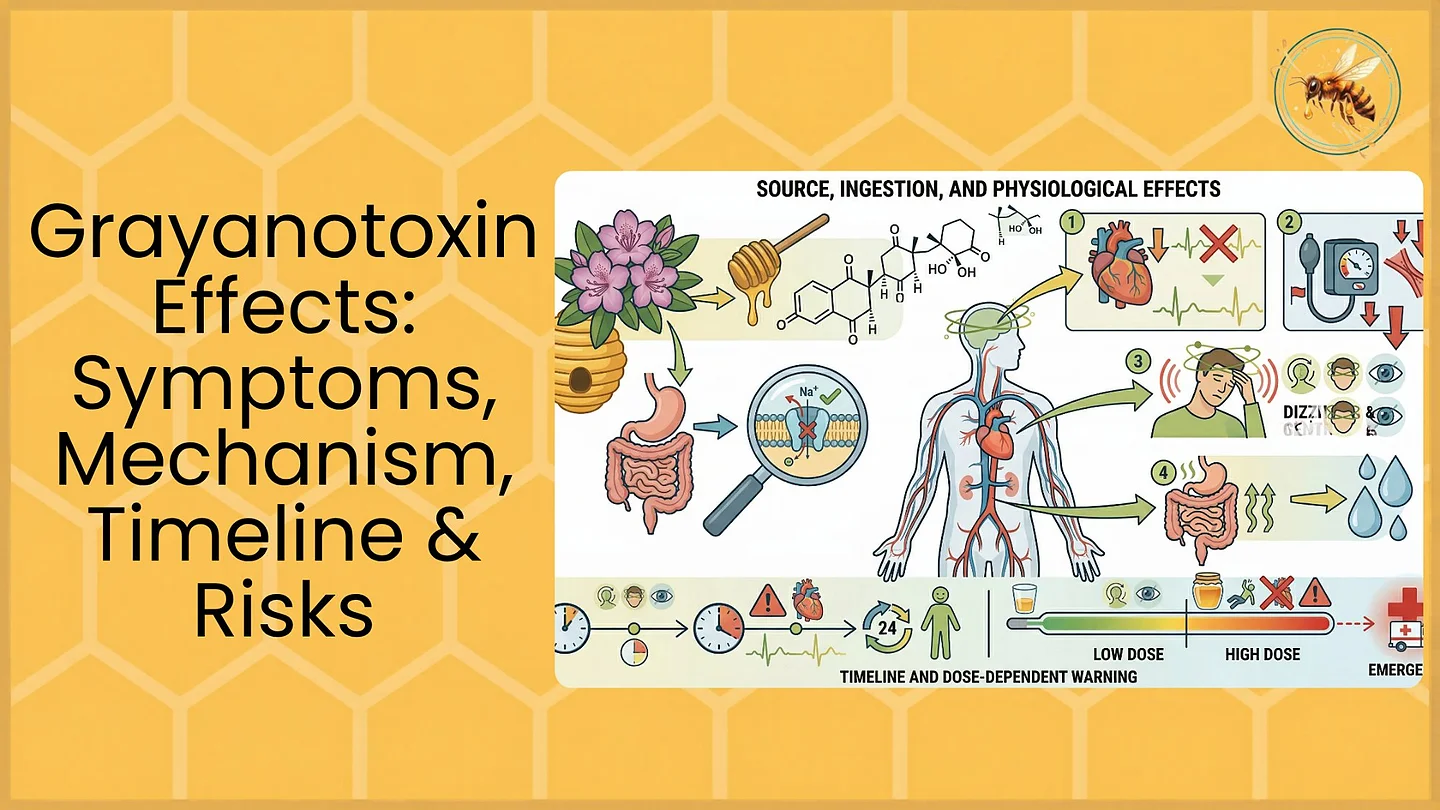
The Mechanism: Why Mad Honey Has Noticeable Effects
To understand why mad honey has a reputation at all, you need one concept: rhododendron nectar can contain grayanotoxins, and those compounds can affect how excitable certain cells are, especially in ways that show up as cardiovascular and “body-first” symptoms.
Rhododendron nectar → grayanotoxins
Mad honey is typically associated with nectar from certain rhododendron species. Grayanotoxins are naturally occurring compounds in some of these plants. When they end up in honey, they can produce physiological effects that are very different from ordinary honey.
Why effects scale sharply with dose
A defining feature of this topic is dose sensitivity. At low amounts, some people report subtle effects; at higher amounts, the experience can shift quickly into uncomfortable or dangerous territory. Dizziness, weakness, nausea, low blood pressure symptoms, and slow heart rate patterns are described in clinical literature. This is one reason the internet feels chaotic: a “small taste” and “several spoonfuls” can lead to completely different outcomes.
Why two people can react very differently
Even if two people take “the same amount,” their responses can differ because bodies differ (baseline blood pressure sensitivity, hydration, sleep, medications, etc.). Then add the second layer: batches vary. If you want a clean mental model, think of mad honey as a product with two variables that stack: human variability plus product variability.
Human Evidence: What Case Reports Typically Describe
This section stays factual and safety-forward because the most reliable human literature is about adverse events and clinical presentation, not wellness outcomes. That doesn’t mean every use leads to harm; it means the papers we have are disproportionately about the moments when something went wrong enough to require medical attention.
Common symptom pattern
Across many reports, a recognizable cluster shows up repeatedly: dizziness or lightheadedness, weakness, nausea and vomiting, sweating, and signs consistent with low blood pressure and slow heart rate in some cases.
People sometimes describe it as feeling “drunk” or “collapsed,” which matters because it explains why “psychedelic” framing is often misleading: the dominant pattern is frequently physical impairment, not a purely mental altered-state.
Typical time course (onset → peak → recovery)
Case literature often notes that symptoms appear after consumption, intensify over a period, and then resolve with time and supportive monitoring. That’s why timeline questions are so common, and why conservative guidance emphasizes waiting and not stacking.
What clinical management commonly involves
Clinical write-ups commonly describe monitoring of vitals, observation, and supportive care approaches in a medical setting. This is not a DIY instruction manual; the point is to set expectations: if someone presents with significant symptoms, clinicians focus on stability and monitoring because the key risk is cardiovascular instability and dehydration from vomiting, not “detoxing” or gimmicky cures.
What Research Does NOT Prove
A science-forward page earns trust by being explicit about the gaps. Mad honey content online often jumps from “there is research” to “there are benefits,” but those are different claims.
“Benefits” vs evidence quality
The word “benefits” implies controlled demonstration: consistent positive outcomes, measured endpoints, and reliable dosing. That’s not what most human mad honey literature provides. What it does provide is a clear warning: effects are real, dose-dependent, and can become adverse.
Anecdotes ≠ clinical trials
Anecdotes can be interesting and culturally meaningful, but they’re not evidence that a product treats or prevents disease. People can report feeling calm, sleepy, or “different,” but that isn’t the same as demonstrating a therapeutic effect in a controlled study.
Why “stronger is better” isn’t supported
In the clinical literature, “stronger” tends to correlate with more symptoms and more risk. The idea that you should chase intensity is the opposite of what safety patterns suggest. If you want to build long-term trust, the durable message is: more isn’t better, more is less predictable.
Why the same jar can’t be assumed consistent
Even within the same product type, batch variability changes the experience and the risk profile. Without standardization, you can’t responsibly generalize from one jar to another, especially not when online listings provide vague origin claims and no meaningful testing transparency.
Animal and Lab Studies: What They Suggest, What They Can’t Confirm
If human literature mostly shows adverse event patterns, why do animal and lab studies matter? Because they help answer “how could this work?” at a mechanistic level. But they have limits, and those limits should be explicit.
What animal studies typically measure
Animal studies often look at cardiovascular markers, behavior, inflammation, or oxidative stress markers, and dose-response behavior, depending on the design. Sometimes the focus is on isolated compounds or extracts rather than real-world honey consumption.
Translating animal results to humans: the big limitations
The first limitation is dose equivalency. A dose that produces a measurable effect in a rat model may not map meaningfully to a human spoonful, especially when the compound concentration isn’t standardized.
The second limitation is the form: studies might use extracts or isolated components, while consumers use whole honey with variable composition. The third limitation is endpoint relevance: changes in biomarkers don’t necessarily mean a real-world health benefit.
Dose equivalency problems + extract vs whole honey differences
This is where many online “science-backed” claims fall apart. If a study uses an extract at a high dose relative to body weight, you can’t ethically or logically convert that into “therefore this jar will do X.” The best way to use these studies is as hypothesis generators: they help point to pathways worth investigating, not conclusions to market as certainties.
Testing and Standardization: The Missing Piece in Most Online Claims
If you only take one technical idea from this article, make it this: testing isn’t just about proving something is “safe,” it’s about making claims meaningful. Without standardization and transparent reporting, “lab tested” becomes a vague marketing phrase.
Measuring grayanotoxins (HPLC/LC-MS overview)
When labs measure grayanotoxins, they’re typically using analytical chemistry methods designed to detect and quantify small molecules. High-performance liquid chromatography (HPLC) is commonly used to separate compounds, and mass spectrometry (LC-MS) can help identify them based on mass/charge signatures and fragmentation patterns.
In plain English: these methods help answer, “Are grayanotoxins present, and roughly how much?” That’s foundational for any meaningful discussion about batch-to-batch variability and why dosing guidance must remain conservative.
Contaminant testing and adulteration risk
Separate from grayanotoxins, honey in general can face contamination and adulteration issues, things like added sugars, mislabeling, or other purity problems. That means a responsible product conversation includes both: “What’s naturally in it?” and “What shouldn’t be in it?” The second question matters even more in marketplaces where traceability and documentation are weak.
What a “responsible batch” should show
A reader doesn’t need lab credentials to spot the difference between transparency and vibes. A responsible seller typically provides clarity on origin, harvest/region context, and batch identification, and if they reference testing, they should be specific about what was tested, what method was used, and how results are linked to that batch.
Practical Safety Takeaways From Research
Science is only useful if it changes behavior in the real world. The consistent message across clinical patterns and mechanisms is not “take more” or “chase effects.” It’s “keep it conservative and predictable.”
Start low, wait, don’t stack
If effects can be delayed, especially with food, stacking becomes the fastest route to turning a mild experience into a problem. Waiting long enough for the onset and peak to declare themselves is a simple behavior that dramatically reduces risk.
Who should avoid it (risk groups)
People with cardiovascular concerns, people who are pregnant, and anyone taking medications that affect blood pressure or heart rhythm should be extra cautious, and in many cases should avoid experimentation altogether. Even without making medical claims, a safety-first page should acknowledge that baseline risk varies, and some groups have less buffer.
Red flags when to seek help
The research-consistent red flags are the ones that show up repeatedly in clinical presentations: fainting or near-fainting, severe weakness, persistent vomiting, chest pain, breathing difficulty, or symptoms that worsen instead of improve. If readers want one rule, it’s trend-based: mild effects should fade; worsening symptoms are not “part of the ride.”
Future Research Questions
If you’re building a science pillar, it helps to show what “better evidence” would look like. This invites citations, academic interest, and external links without overselling certainty.
Better dose-response mapping
The most practical research gap is establishing clearer dose-response curves that reflect real-world consumption, not just isolated compound models. That means observational data that connects approximate intake with symptom severity and duration.
Standardized batch reporting
A major advance would be standardized reporting of grayanotoxin ranges per batch (with clear methods and limitations), allowing consumers and researchers to compare apples to apples.
Controlled observational studies
Randomized trials may be hard, but well-designed observational studies could still improve understanding: what doses people take, what effects they report, which risk factors correlate with adverse events, and how long symptoms typically last.
Regional comparisons (Nepal vs Turkey) with chemistry profiles
There’s a public interest in regional differences, but it’s often handled as marketing lore. Comparative studies that combine origin verification with chemistry profiles would improve the quality of conversation dramatically.
Conclusion
If you strip away hype and treat the evidence honestly, the mad honey research story becomes clear: the strongest evidence supports mechanism and safety risk patterns, not dramatic benefit claims.
Human literature is dominated by case reports describing dose-dependent adverse effects and typical recovery timelines. Animal and lab studies can suggest pathways, but can’t confirm human outcomes without standardized dosing and well-designed human observational work.
The most research-aligned takeaway for readers is practical: set conservative expectations, choose transparency over marketing, and treat dosing as the control knob, start low, wait, don’t stack, and take red-flag symptoms seriously.
FAQs on Mad Honey Research
Is there scientific proof mad honey “works” for X?
For most “benefit” claims, the evidence isn’t strong enough to say it works in a clinical sense, and it’s safer to treat those claims as unproven, especially when they drift into medical territory.
Why do most papers discuss poisoning?
Because the most common human publications are clinical reports written when something went wrong. That literature is still valuable because it reveals repeatable patterns and real safety constraints.
Is mad honey considered safe in studies?
The research doesn’t support a blanket “safe” label because outcomes depend heavily on dose, sensitivity, and batch variability; the most defensible stance is conservative use with clear risk awareness.
What’s the role of grayanotoxins?
They’re the key mechanism discussion because they plausibly explain why effects are noticeable and why adverse cardiovascular-type patterns appear in clinical reports.
Why does mad honey vary by batch?
Because nectar sources, seasons, regions, and handling vary, which can change compound composition and intensity from jar to jar.
What should a lab test include?
At minimum, testing that is batch-linked, method-specific, and transparent about what was measured (and what wasn’t), with clear documentation rather than vague “lab tested” language.